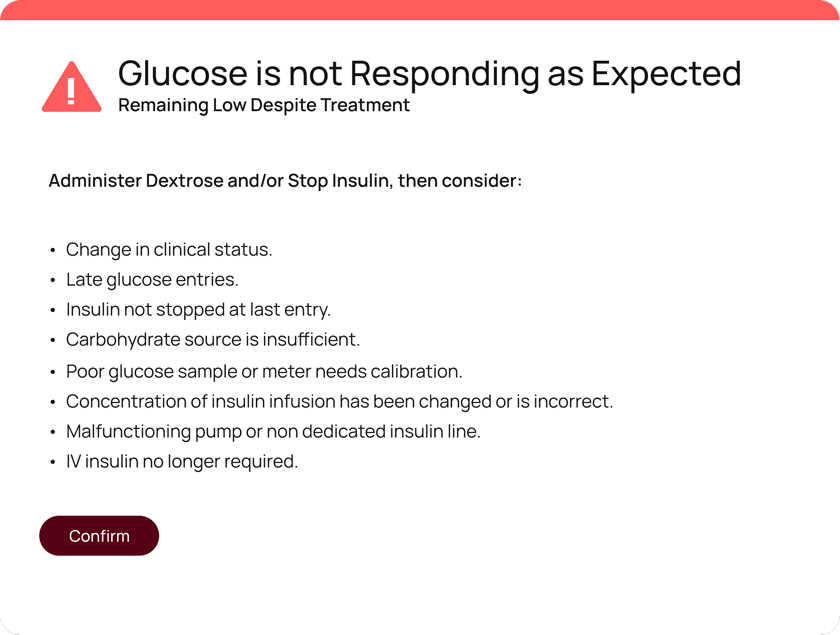
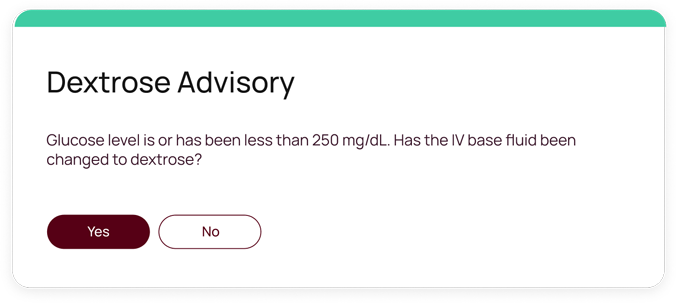
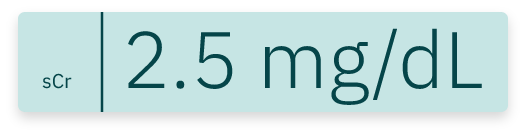Developed by Monarch Medical Technologies, a Glooko Company, EndoTool is the only patient-specific insulin dosing system which simplifies the complex task of glycemic management in hospitals environments.
Unique algorithm
Reduce hypoglycemia
Enhance your workflow
Our system simplifies the complexity and cognitive burden of insulin dosing, allowing healthcare teams to allocate more time to direct patient care.
Standardize care delivery
EndoTool standardizes insulin dosing protocols, reducing variability in care and ensuring consistent treatment practices across your team.
Safe and simple patient-specific insulin dosing, enabled by technology.
Get in Touch
Ready to get started?
"*" indicates required fields
Unmatched safety and effectiveness
What sets EndoTool apart?
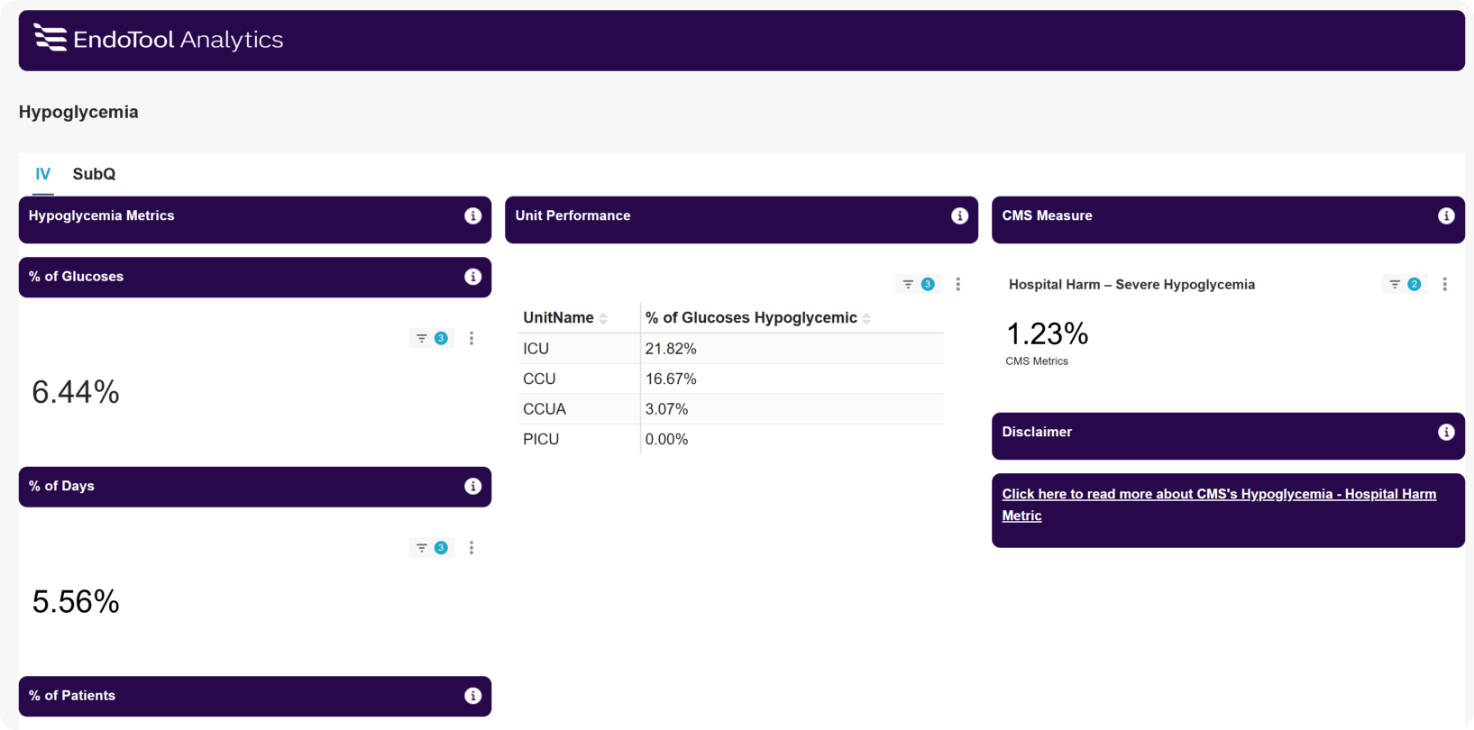
EndoTool precisely calculates residual insulin levels to prevent excessive insulin administration, significantly reducing the risk of hypoglycemia. This is especially effective in patients with kidney failure or reduced renal function, where insulin has a prolonged half-life.
EndoTool’s advanced Estimated Residual Extracellular Insulin (EREI) calculation offers unmatched safety and effectiveness, making it the most reliable option for precise insulin dosing.
How does EndoTool compare?







EndoTool
Multifactorial Model
Competitor
Variable Multiplier
Hospitals using EndoTool consistently improve operations
A direct path to quality care
Real world results

Case Study
Atrium Health’s Use of EndoTool IV Leads to $12 Million in Savings

Customer Stories
Improved Nurse Confidence at Sharp HealthCare Drives Hypoglycemia Down to 0.005%
Case Study
How Temple Health Reduced Hypoglycemia by Over 70% with EndoTool SubQ
Customer Stories
Why CaroMont Health Made EndoTool a Systemwide Standard for Insulin Dosing

Case Study
Corewell Health Slashes DKA Costs by Nearly $1M in 18 Months

Customer Stories
Why Cone Health Replaced their Insulin Dosing Software with Patient-Specific Dosing
Trusted by the best
Discover why leading hospitals choose EndoTool

Trinity Health’s 18-Year Journey with EndoTool®

Tracey Melhuish, RN, MSN, CCRN
Clinical Practice Specialist Critical Care

Beacon Health System’s Successful Journey to Zero Hypo

Kim Henke
RN, MSN, MPA, CCRN-CSC CNS for Critical Care Services

Integrating EndoTool into Northside’s existing systems and workflows was a concerted effort involving critical care teams, clinicians, pharmacy staff, IT professionals, diabetes educators, and providers.

Dr James S. Lee, MD
Physician Lead Inpatient Internal Medicine, Northside Hospital Gwinnet